 ARDSĪRDS, or Acute Respiratory Distress Syndrome, is an inflammatory lung condition involving both lungs that may complicate severe pneumonia (including influenza), trauma, sepsis, aspiration of gastric contents, and many other conditions. The NHLBI-funded Prevention and Treatment of Acute Lung Injury (PETAL) Network will begin work on prevention and early treatment of ARDS. 
The clinical trial data and remaining biospecimens for all but the most recent ARDS Network trial (SAILS) are available to the public through BioLINCC ( ) and the SAILS data and specimens will be available in early 2015. On June 30th, 2014, the Network contract came to a close. Finally, through the foresight of many investigators within the Network, additional resources were obtained for the collection and analysis of biospecimens and to perform numerous ancillary studies, efforts that have advanced our understanding of the pathogenesis and natural history of ARDS. Comparative effectiveness trials using explicit methodology allowed other research groups to replicate and build upon the Network’s studies. The ARDS Network also developed new outcome measures (ventilator free days) and promoted innovative and efficient techniques (factorial designs and coenrollment) to speed the discovery of new treatment approaches for patients with ARDS. Additional trials informed best practices by suggesting no role for routine use of corticosteroids, beta agonists, pulmonary artery catheterization, or early full calorie enteral nutrition. In some of the most highly cited articles in critical care, network investigators reported improved survival with lung protective ventilation and shortened duration of mechanical ventilation with conservative fluid management. During its 20 years of service, 5,527 patients were enrolled in 10 randomized controlled trials and one observational study The goal of the Network was to efficiently test promising agents, devices, or management strategies to improve the care of patients with ARDS. The ARDS Network was established as a contract program in 1994 and renewed in 2005 following two national competitions. In order to hasten the development of effective therapy for Acute Respiratory Distress Syndrome (ARDS), the National Heart, Lung, and Blood Institute (NHLBI), National Institutes of Health (NIH), initiated a clinical network to carry out multi-center clinical trials of ARDS treatments. 
The safe range of ventilation for this patient is 315-420ml and the respective tidal volumes for 6, 7, and 8 ml/kg are 315, 365 and 420 ml respectively.The NIH-NHLBI ARDS Network was a research network formed to study treatment of Acute Respiratory Distress Syndrome. Therefore, the safe range of ventilation is 490-660ml (6-8ml/kg) and the respective tidal volumes for 6,7,8 ml/kg are 490, 575 and 660 ml respectively. Tidal volume of 8ml/kg = 82.2 kg x 8 ml/kg Tidal volume of 7ml/kg = 82.2 kg x 7 ml/kg Tidal volume of 6ml/kg = 82.2 kg x 6 ml/kg Remember, the safe range for tidal volumes is 6-8 ml/kg. We have already learned how to calculate IBW (steps 1 and 2), so let’s try step 3 using the IBWs that were calculated earlier in this chapter. Note: you will always round your result up or down to match the settings on the ventilator (usually, they use whole numbers only and count by 5s). Multiply your 6, 7 and 8 ml/kg and write these in your chart, so you have a handy reference point for the tidal volumes you can use now and in the future.Use appropriate IBW formula (male versus female) to calculate your IBW.Determine your patient’s height and sex. 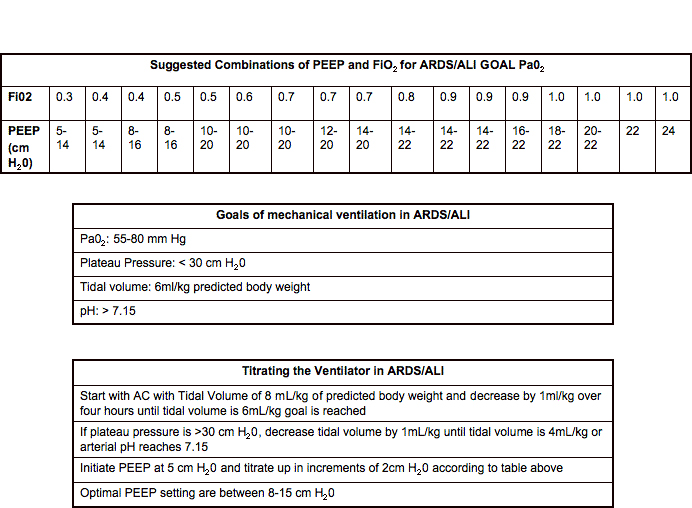
Let’s review the steps to determine a safe tidal volume for your patients. Once the IBW is calculated, it is multiplied by 6 and then 8 to get the range of safe tidal volumes for a patient on a mechanical ventilator. Today, 8 ml/kg is the hard limit for tidal volumes to be set by clinicians in control modes. This discovery revolutionized tidal volume strategies and has become the gold standard of adult tidal volume ranges to decrease the risk of VILI from volutrauma/barotrauma. The results were conclusive that using tidal volumes of 6-8 ml/kg of ideal body weight directly improved morbidity and mortality. Researchers also studied patients diagnosed with ARDS to see if the amount of tidal volume impacted morbidity and mortality. In the early 2000s, many studies were done to find the best tidal volume range based on ideal body weight to decrease the risk of VILI and ARDS. This approach still resulted in volutrauma and VILI. Even with the determination of using the ideal body weight instead of actual body weight, historically, tidal volumes of 10 ml for every kilogram of IBW were often used. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
